There is market pressure for a PRP product with decreased red blood cell contamination, especially in aesthetic and cosmetic procedures and in sports medicine to reduce potential complications in joint treatment. Reduced granulocyte levels may also be desirable.
While granulocytes are helpful in wound debridement and preventing infection, high granulocyte levels may be inflammatory. You can also get more information about emcyte suppliers in NY through various sources.
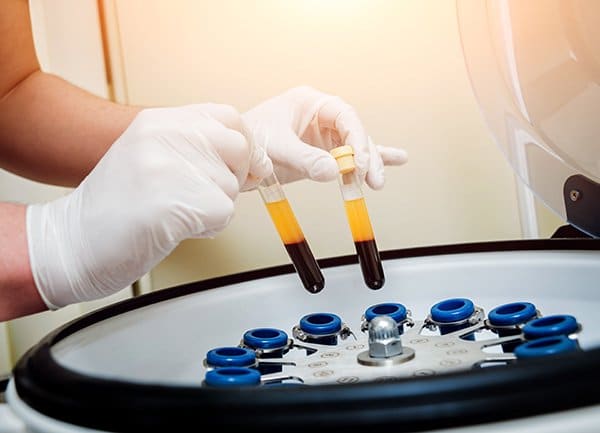
Image Source: Google
PRP is the leader in platelet-rich plasma systems. With over 800,000 patient cases worldwide since 2013, Eclipse is recognized as the trusted leader in Platelet-Rich Plasma technology. More of the top PRP medical providers use and recommend Eclipse PRP® over any other brand.
This study assessed the PRP products from platforms such as Emcyte PRP in NY, these are red cell reduction methods and have a programmable setting to control the RBC level in the product. The study is a paired sample design, with each donor tested on all the PRP platforms.
The Emcyte PRP is an advanced centrifuge system that delivers the highest amount of deliverable platelets in only 6 minutes with a hematocrit of less than 1%. This allows for smaller gauge needles. The centrifuge is sleek, digital, self-balancing, and FDA-cleared.
The Pure PRP device produced a reduced Red Blood Cells PRP product. Only 2% of the granulocytes were retained, a reduction of 84% from the baseline whole blood values. one of the most, if not the most, flexible PRP systems on the market. For more information about emcyte suppliers in NY, you can search the web.

Image Source: Google
The Pure PRP products had higher cell concentration and calculated cell metrics including platelet yield and concentration, RBC, mononuclear, and granulocyte cell recoveries than either the clear PRP.
The average concentrations for all growth factors measured were higher in Pure PRP products compared to others; however, The difference was not significantly different between the Pure PRP and Clear PRP products. Both red cell reduction platforms had similar processing times and the number of aseptic entries.
